What is in the STEM-PD product and how does it work?

The STEM-PD product is a solution of dopamine progenitor cells, which means that they are immature cells which can form functional dopamine neurons upon maturation in the brain. To obtain maximal clinical benefit, the cells are implanted into the area of the brain where dopamine normally exerts its effects on motor functions, which is a region called the putamen. The STEM-PD product is derived from stem cells which have the ability to both, multiply (self-renew) and mature (differentiate) into any specialised cell type in the body. In the laboratory stem cells can be differentiated into dopamine progenitor cells, parent cells of the dopamine neurons whose loss results in the symptoms of PD.
In the STEM-PD trial, the clinical team will transplant dopamine progenitor cells into the brains of people with PD, placing them within the main area of the brain where dopamine normally exerts its effects on motor functions. The goal is that the progenitor cells will mature into dopamine neurons and release dopamine into the brain in a more natural way.
This type of therapy is particularly suited for PD because the cell type and brain area that needs repair are well defined and the number of cells required is relatively few.
To learn more about the technology behind the STEM-PD cells, see the information below.

What are the STEM-PD cells?
The STEM-PD cells are dopamine progenitor cells which are derived from a special type of stem cells called pluripotent stem cells. As the original pluripotent stem cells can divide and multiply, they can generate a large number of dopamine progenitors which can be stored (cryo-preserved) for a long time, thus making it possible to do extensive testing of the exact same cells that will be used in clinical trial participants. The preclinical testing which has been performed on STEM-PD includes assessing both the safety and the efficacy of the cells in animal models. In the STEM-PD trial, the cryopreserved dopamine progenitor cells will be thawed and transplanted into both sides of participants’ brains during one surgery. After the transplantation, the progenitor cells will mature into dopamine neurons that produce dopamine. The process of maturation takes many months, and based on similar trials using fetal tissue, it may take 1-3 years until the full effect of the STEM-PD cells is seen.
What are pluripotent stem cells?
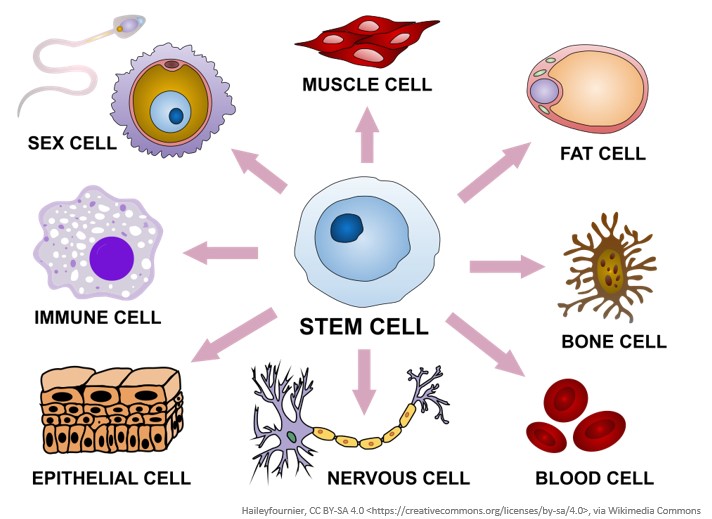
Pluripotent stem cells have unique characteristics in that they have the ability to both multiply (self-renewal) and mature (differentiate) into any specialised cell type in the body. The term pluripotent means that these stem cells have the potential to make any cell type as they differentiate.
Stem cells are able to change and transform (differentiate) into other types of cells found in the body. This means that stem cells can differentiate into muscle cells, fat cells, bone cells, blood cells, nervous cells, epithelial cells, immune cells, sex cells, and more.
The human pluripotent stem cells used for generating the STEM-PD product come from human embryonic stem cells, grown in the laboratory from a surplus embryo from in vitro fertilisation.
The unique characteristics of pluripotent stem cells make them highly valuable for medical research, drug discovery and regenerative medicine. As the stem cells can multiply and provide an unlimited supply of human cells for research purposes, scientists are able to address many questions in the laboratory before moving to clinical trials.
How can pluripotent stem cells be used to make dopamine neurons?
The main focus of the STEM-PD team, and many colleagues around the world, has been to better understand how human dopamine neurons develop, and how stem cells can be made to mature into midbrain dopamine neurons in the laboratory, for potential regenerative medicine applications. This collected body of work has enabled the development of technologies that can be used to guide the maturation of human pluripotent stem cells into dopamine progenitors at a high efficiency over a 16-day period. This has allowed for the manufacturing of STEM-PD. Although these stem cell-derived dopamine cells are produced in the lab, they are molecularly and functionally indistinguishable from the dopamine cells found in the human brain. Consistently, when the STEM-PD cells have been transplanted to the rodent brain during preclinical testing, the cells have matured into functional dopamine-producing neurons which were able to reverse motor symptoms in animal models of PD.
How do we know that the STEM-PD cells are safe and effective?
The STEM-PD trial will be the first time the STEM-PD cells are tested in humans. Prior to this, the cells have undergone very rigorous preclinical safety and efficacy testing in animal models. This testing has been reviewed and approved by the Ethical review board and the Swedish Medical Products Agency (MPA) who are responsible for regulating medicines in Sweden. For the patients coming from UK, a similar approval from the Medicines and Healthcare Prodicts Regulatory Agency (MHRA) will be sought.
To ensure the highest level of safety, the scientists behind the STEM-PD product have performed rigorous preclinical experiments using the exact STEM-PD cells that will be transplanted to participants in the clinical trial. The key findings from these preclinical studies are:

01
Cell Survival
The transplanted STEM-PD cells can survive long-term in the brains of rats, where they mature into dopamine-producing neurons making long-range connections with the host brain neurons.
02
Symptom Reversal
The transplanted STEM-PD cells can reverse motor deficits in animal models of PD, indicating a potential to ameliorate dopamine-related motoric symptoms in patients.
03
Safety
Large safety studies in rats with STEM-PD cells have shown that the cells are safe; they maintained long-term within the site of transplantation with no signs of distribution to other sites in the body; no signs of toxicity; and no tumour formation.
